The FDA has given its approval for DermaSensor’s AI-powered handheld skin cancer detector to be sold in the US.
Skin cancer is the most common cancer in the world with around one in five Americans developing skin cancer in their lifetime. The most serious form of skin cancer, melanoma, kills about 9,000 Americans each year.
About 99% of skin cancers are curable, but they often go undetected until it’s too late. Dermatologists are in short supply and it can be difficult for a primary care physician (PCP) to tell the difference between a benign mole and skin cancer.
DermaSensor designed a non-invasive handheld device that can detect the three most common forms of skin cancer, melanoma, basal cell carcinoma, and squamous cell carcinoma.
The device uses elastic-scattering spectroscopy (ESS), a noninvasive optical biopsy technique. When the tip of the device is held against the suspect skin lesion, it pulses multiple wavelengths of light through it and captures the resulting spectra.
It then uses an algorithm to analyze the spectra to determine if the lesion is malignant or benign.
https://youtu.be/yUaEgpMj8SY
The algorithm was trained with over 20,000 spectral scans from more than 4,500 skin lesions using one feedforward and two deep neural networks. Once trained on the labeled spectral scans, the device learned to tell if a new spectral scan was cancerous and what type of skin cancer it was.
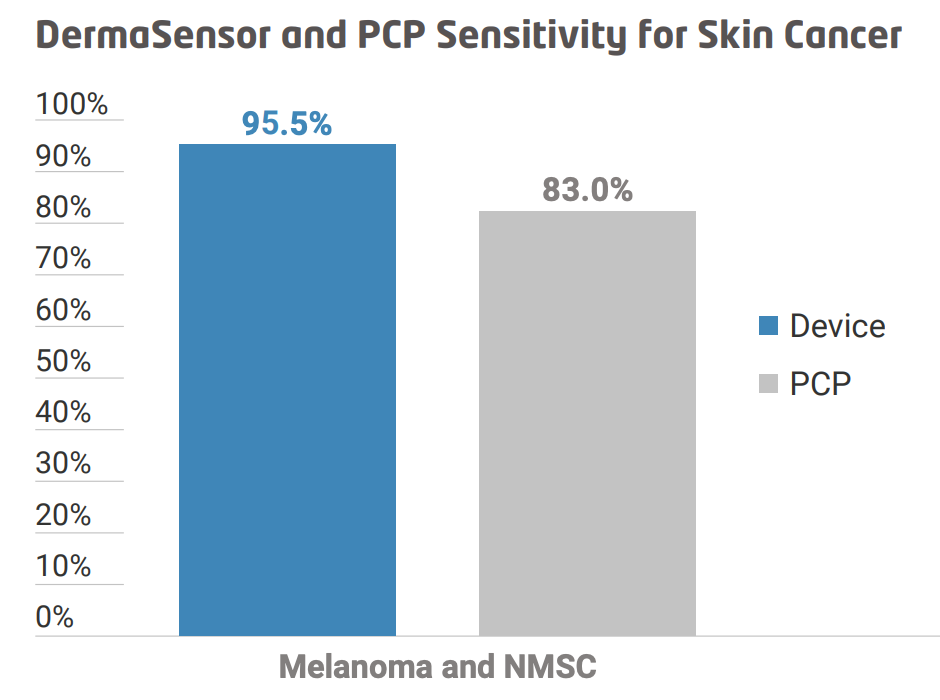
The device positively identifies skin cancer with a 96% accuracy, while it correctly identifies benign lesions 97% of the time. These figures are significantly better than those achieved by the PCPs in their trials.
The device will be made available through a subscription model at $199 a month for five patients or $399 a month for unlimited use.
DermaSensor will enable PCPs to quickly determine whether their patient should be referred to a dermatologist. It will also reduce unnecessary referrals of patients to specialists only to find that the overcautious PCP misidentified a benign lesion.
This is one of several cancer detection and treatment solutions that AI has made possible.





